PG1995
Active Member
Hi
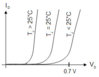
Q1: Silicon has barrier potential of 0.7 V and germanium has barrier potential of 0.3 V. I think barrier potential is dependent upon doping agents used. Please correct me if I'm wrong. The question is why silicon and germanium have different barrier potential.
This is what I think. The amount of energy required to free an electron from the valence band of a silicon atom is called the band-gap energy and is 1.12 eV (electron volts) and for germanium it's almost 0.78 eV. The difference between the band-gap energies shows that in case of silicon electrons are more tightly bound to positively charged nucleus. So, when depletion region has been formed, positive ions on the side of n-region pull less vigorously on the electrons on the side of p-region in case of germanium than for the case of silicon. Do I make sense?
Q2: A solar panel's voltage is negatively affected by temperature - voltage drops with increasing temperature. On the other hand, the panel's current increases with increasing temperature to a small degree. What I want to understand is that why the voltage decreases with increasing temperature.
In answer #2 on this link provides a clue but I'm still struggling to grasp it.
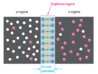
Suppose we have a silicon solar cell as show here but assume that initially there are no electron-hole pairs. There will be a barrier potential of, possibly, 0.7 V. When photons start hitting the solar cell, electron-hole pairs are generated. For the sake of simplicity, we will focus on only one electron-hole pair which is generated in the n-region. As it is mentioned in answer #2 that at elevated temperature it needs less energy to unbound an electron from an atom. How would this translate to less open circuit voltage for a solar panel at elevated temperatures?
Thank you for your help.
Regards
PG
Q1: Silicon has barrier potential of 0.7 V and germanium has barrier potential of 0.3 V. I think barrier potential is dependent upon doping agents used. Please correct me if I'm wrong. The question is why silicon and germanium have different barrier potential.
This is what I think. The amount of energy required to free an electron from the valence band of a silicon atom is called the band-gap energy and is 1.12 eV (electron volts) and for germanium it's almost 0.78 eV. The difference between the band-gap energies shows that in case of silicon electrons are more tightly bound to positively charged nucleus. So, when depletion region has been formed, positive ions on the side of n-region pull less vigorously on the electrons on the side of p-region in case of germanium than for the case of silicon. Do I make sense?
Q2: A solar panel's voltage is negatively affected by temperature - voltage drops with increasing temperature. On the other hand, the panel's current increases with increasing temperature to a small degree. What I want to understand is that why the voltage decreases with increasing temperature.
In answer #2 on this link provides a clue but I'm still struggling to grasp it.
Suppose we have a silicon solar cell as show here but assume that initially there are no electron-hole pairs. There will be a barrier potential of, possibly, 0.7 V. When photons start hitting the solar cell, electron-hole pairs are generated. For the sake of simplicity, we will focus on only one electron-hole pair which is generated in the n-region. As it is mentioned in answer #2 that at elevated temperature it needs less energy to unbound an electron from an atom. How would this translate to less open circuit voltage for a solar panel at elevated temperatures?
Thank you for your help.
Regards
PG